Industry-Specific Solutions
Medical Devices. Packaging.
Perfected.
FDA-compliant medical device packaging
Compliance Information
All sterile packaging validated per ISO 11607-1 and 11607-2. Manufacturing follows FDA 21 CFR 820 Quality System Regulation and QMSR requirements. Materials compatible with EtO, gamma, and e-beam sterilization. UDI labeling per FDA GUDID requirements. EU MDR packaging requirements supported. Cleanroom packaging environments available for Class II and III devices.

Why Packaging Matters for Medical Devices
Medical device packaging operates at the intersection of patient safety, regulatory compliance, and supply chain integrity. Every package must maintain sterile barrier integrity from manufacturing through sterilization, distribution, and point-of-use presentation in clinical settings. Cubit builds custom packaging systems across all medical device classifications. For sterile devices, we produce thermoformed PETG trays with DuPont Tyvek lidstock validated to ISO 11607, chevron peel pouches with sterilization process indicators, and double-barrier systems for high-value implants. For non-sterile devices, we create custom folding cartons with regulatory labeling panels, rigid protective cases with die-cut foam inserts for electronic instruments, and blister cards for disposable components. Our manufacturing follows cGMP protocols with documented process validation for forming, sealing, and assembly. Materials are selected for compatibility with EtO, gamma, and electron beam sterilization methods. Every packaging design includes provisions for UDI compliance, lot tracking, and regulatory labeling in accordance with FDA 21 CFR 820 and EU MDR requirements.
Packaging Solutions for Medical Devices
Explore our specialized packaging options organized by product type.
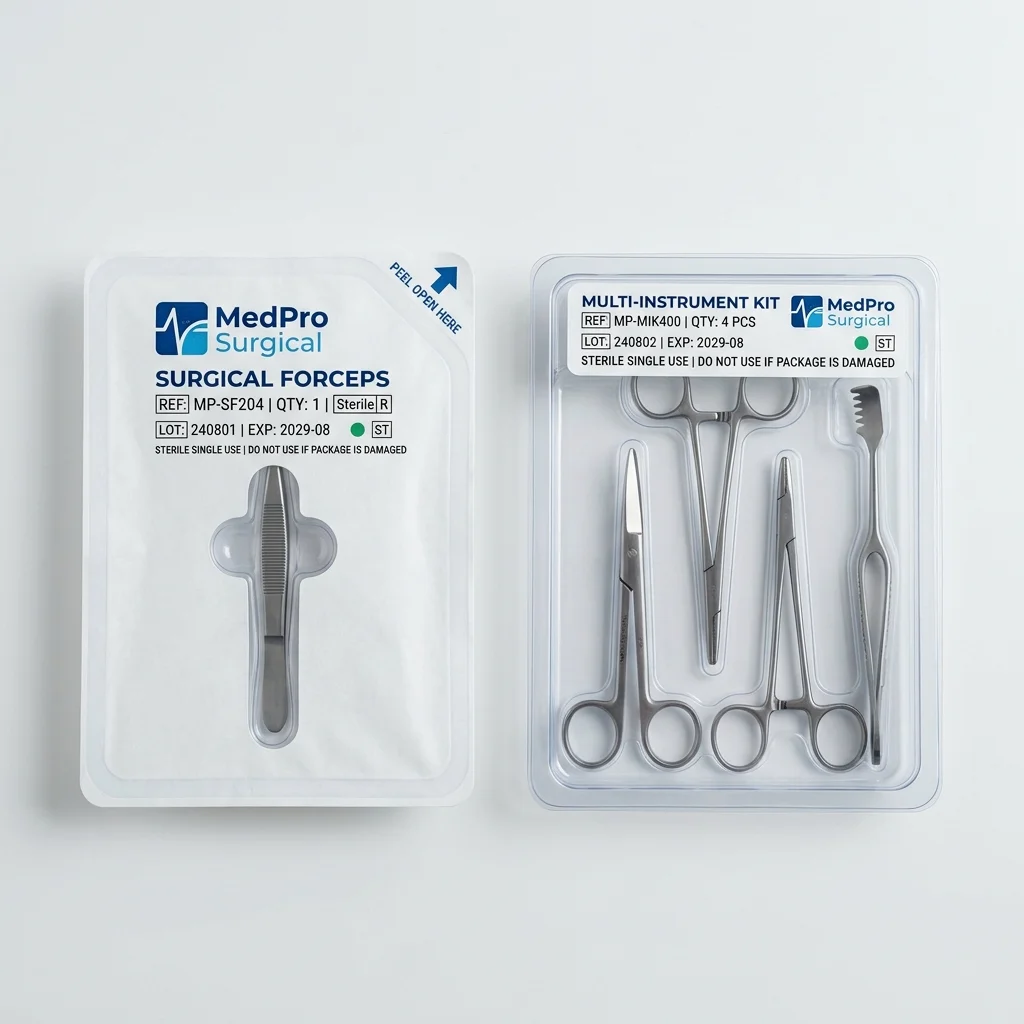
Best for: Thermoformed PETG trays with Tyvek lids, Multi-cavity instrument organizer trays
Surgical Instruments
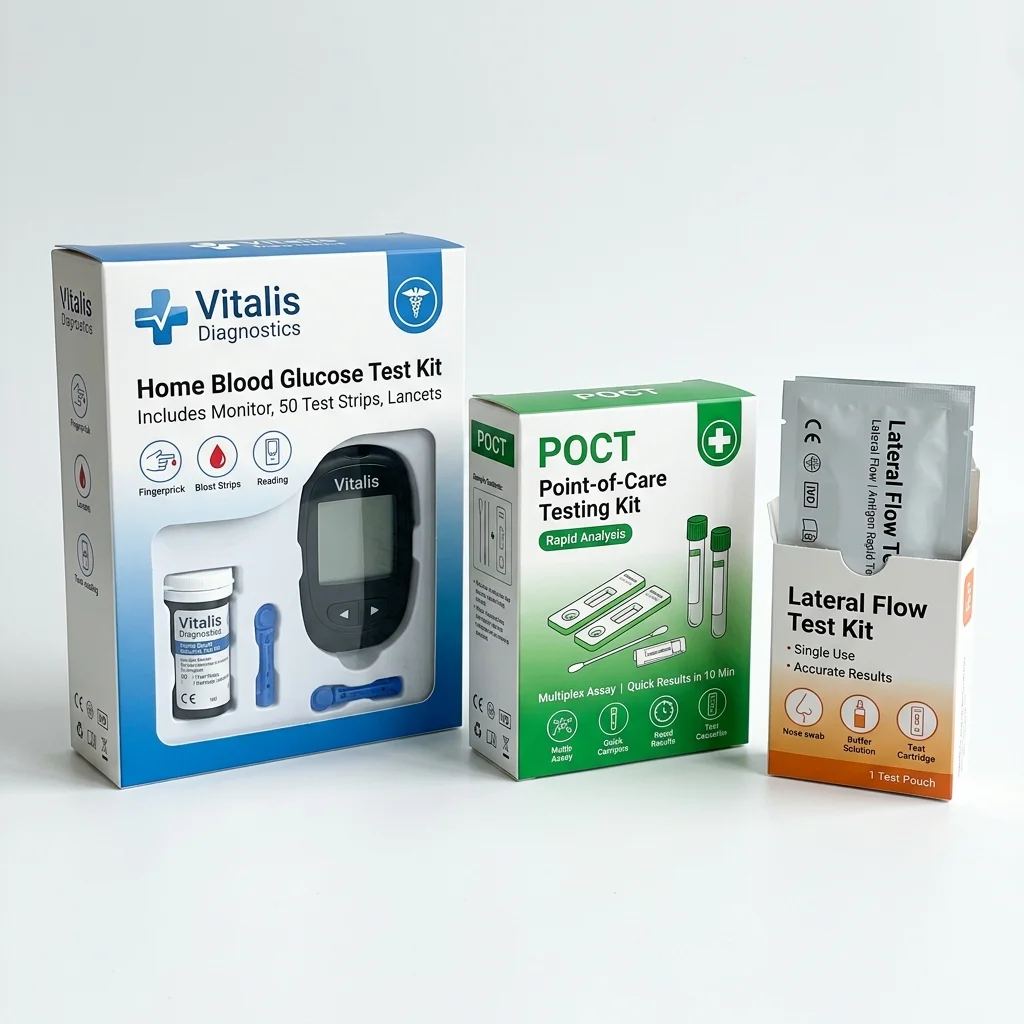
Best for: Printed folding cartons with instructional graphics, Point-of-care test kit packaging
Diagnostic Kits

Best for: High-barrier thermoformed clamshells, Double-pouch sterile barrier systems
Implants and Prosthetics
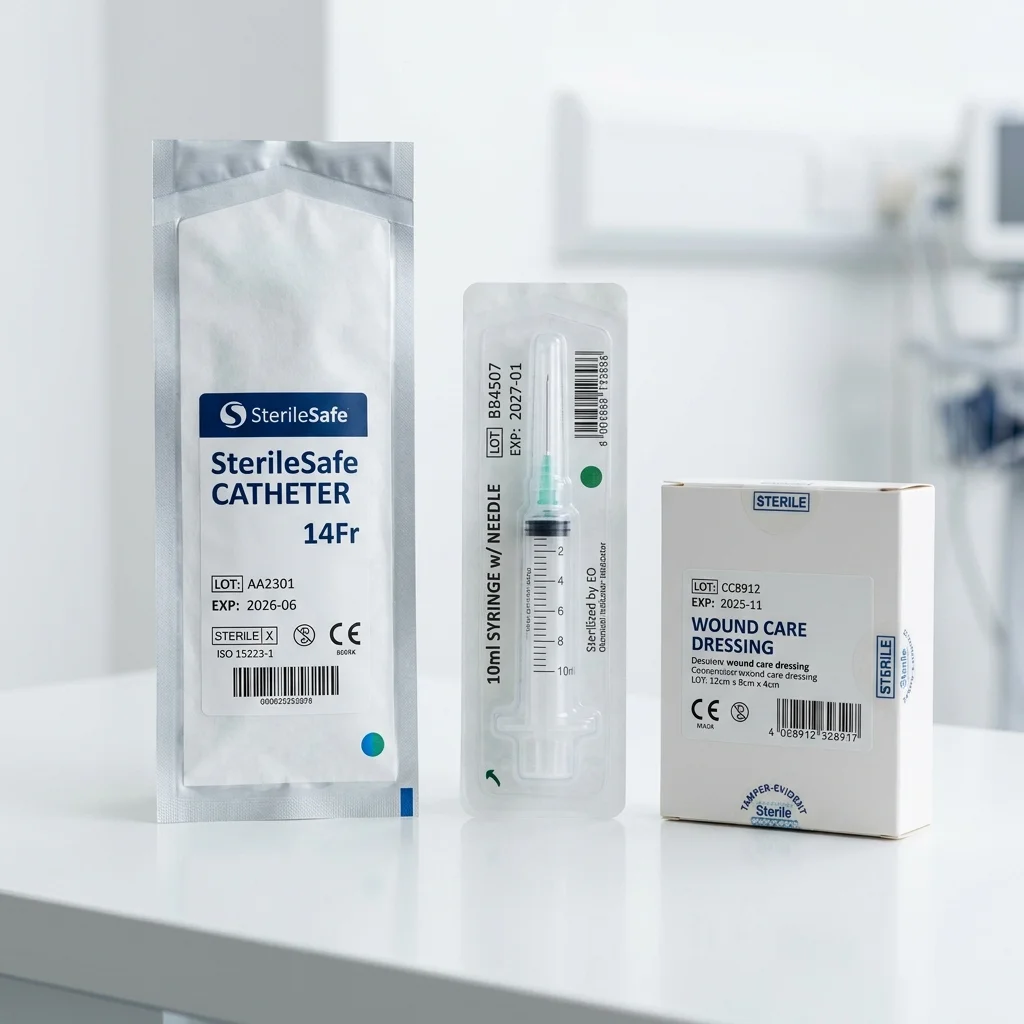
Best for: Chevron peel sterile pouches, Blister card packaging with backing
Single-Use Disposables

Best for: Rigid boxes with die-cut foam inserts, ESD-safe anti-static lined cartons
Electronic Medical Devices

Best for: Insulated corrugated shippers, Gel pack and EPS foam systems
Cold Chain and Temperature-Sensitive
Showing 8 of 20 products

Medical Devices
Rigid Box with Foam Insert
Rigid box with precision-cut foam inside. Absorbs impact and holds products in exact position.

Medical Devices
Tamper-Evident Labels
Labels that show VOID or shatter when peeled. Security verification for pharma, food, and electronics.

Medical Devices
Foam Inserts
CNC-cut or die-cut foam that cradles your product inside the box. Polyethylene, EVA, or polyurethane.

Medical Devices
Tamper-Evident Tape
Tape that leaves a VOID message on the box if removed. Proves the package was not opened in transit.
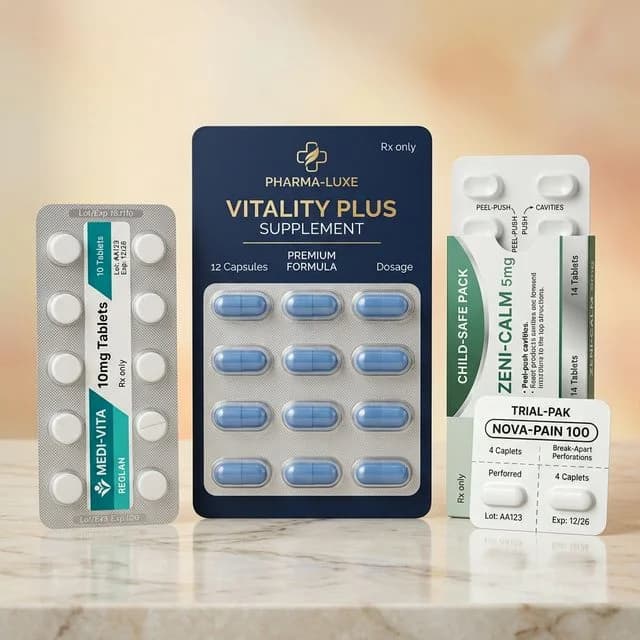
Medical Devices
Blister Packs
Pill and tablet blisters with push-through foil. Pharma-grade, tamper-evident, FDA compliant.

Medical Devices
Custom Folding Cartons
The most versatile box in packaging. Works for pharma, food, beauty, and retail. SBS or kraft board.

Medical Devices
Reverse Tuck End Boxes
Standard folding cartons with opposite-direction tuck flaps. Clean look for cosmetics and OTC pharma.

Medical Devices
Straight Tuck End Boxes
Both flaps tuck the same direction. Simple folding carton for supplements, health products, and electronics.
Top Packaging for Medical Devices
Industry-specific packaging solutions designed for growing brands.
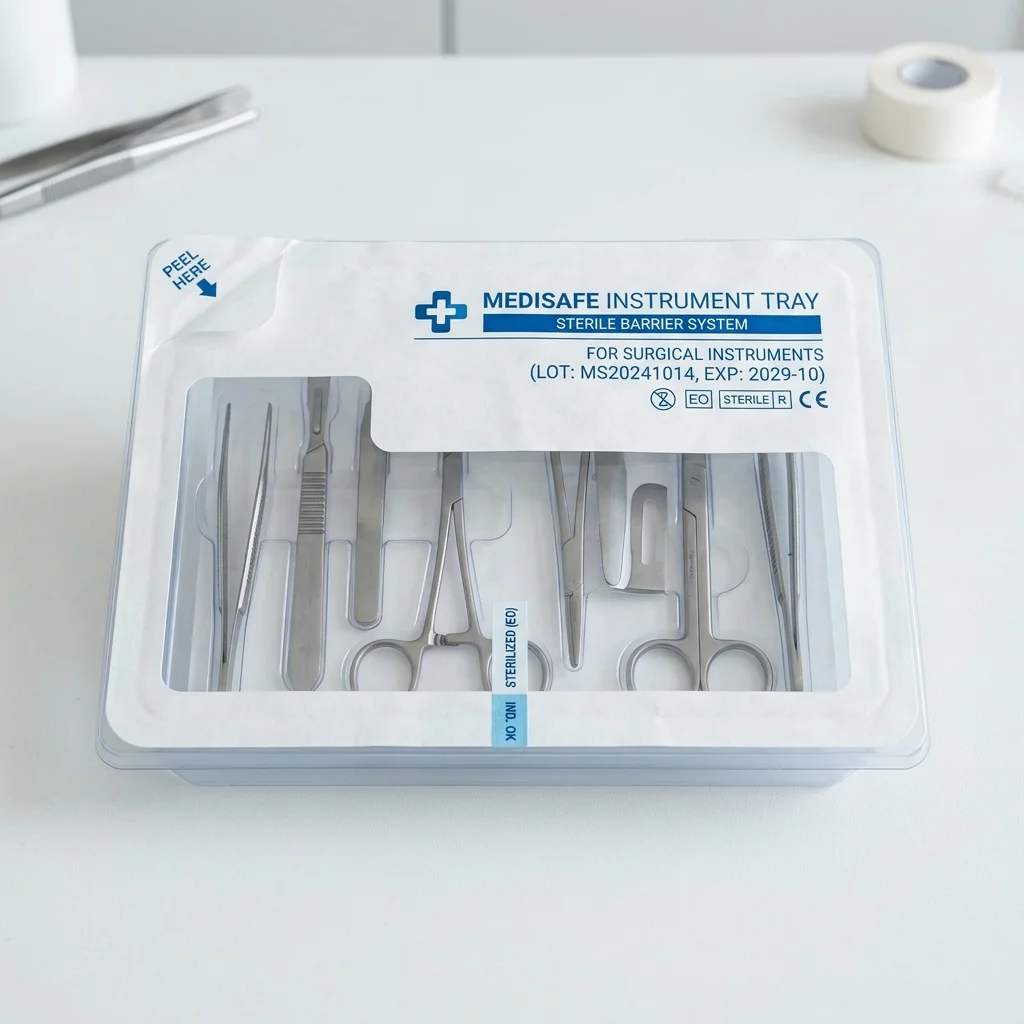
Best for: Surgical instruments and implants requiring terminal sterilization
Sterile Barrier Tray System
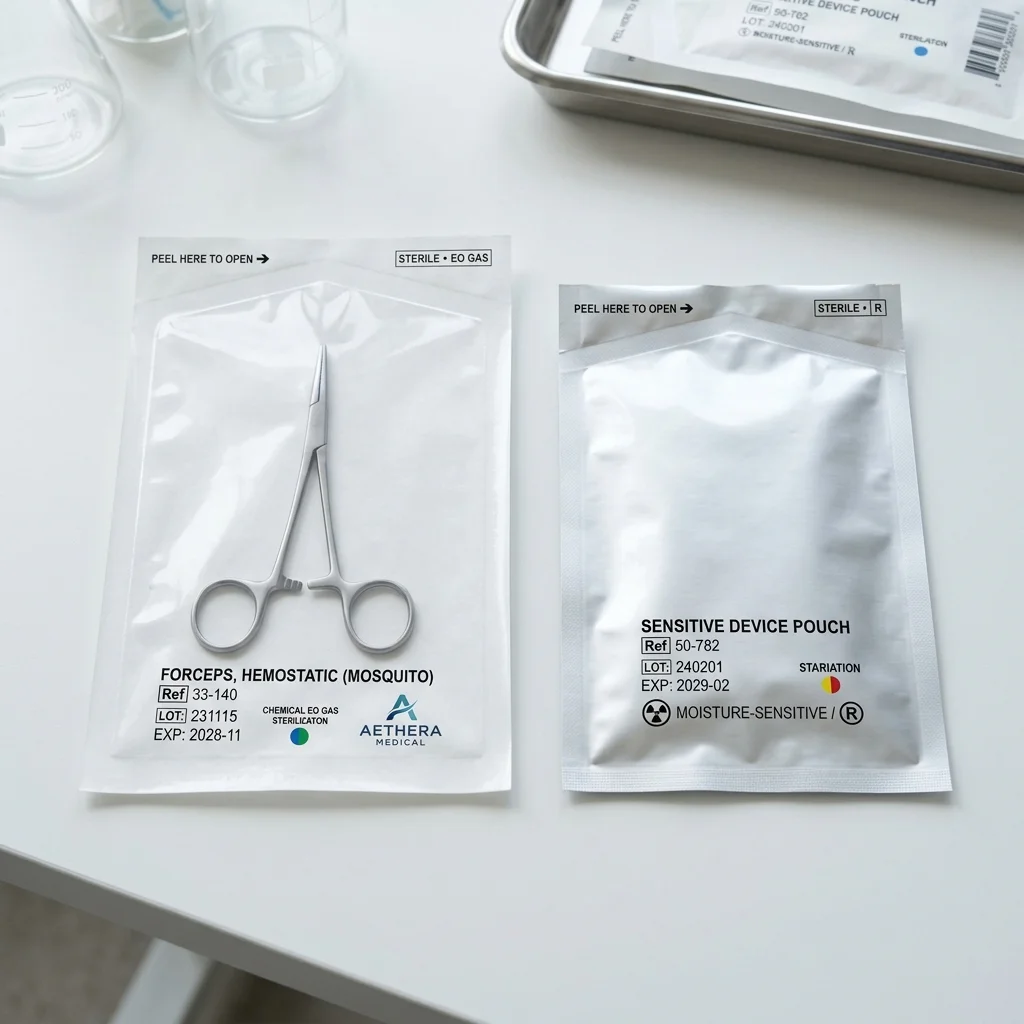
Best for: Diagnostic kits, home-use devices, and retail medical products
Medical Folding Carton

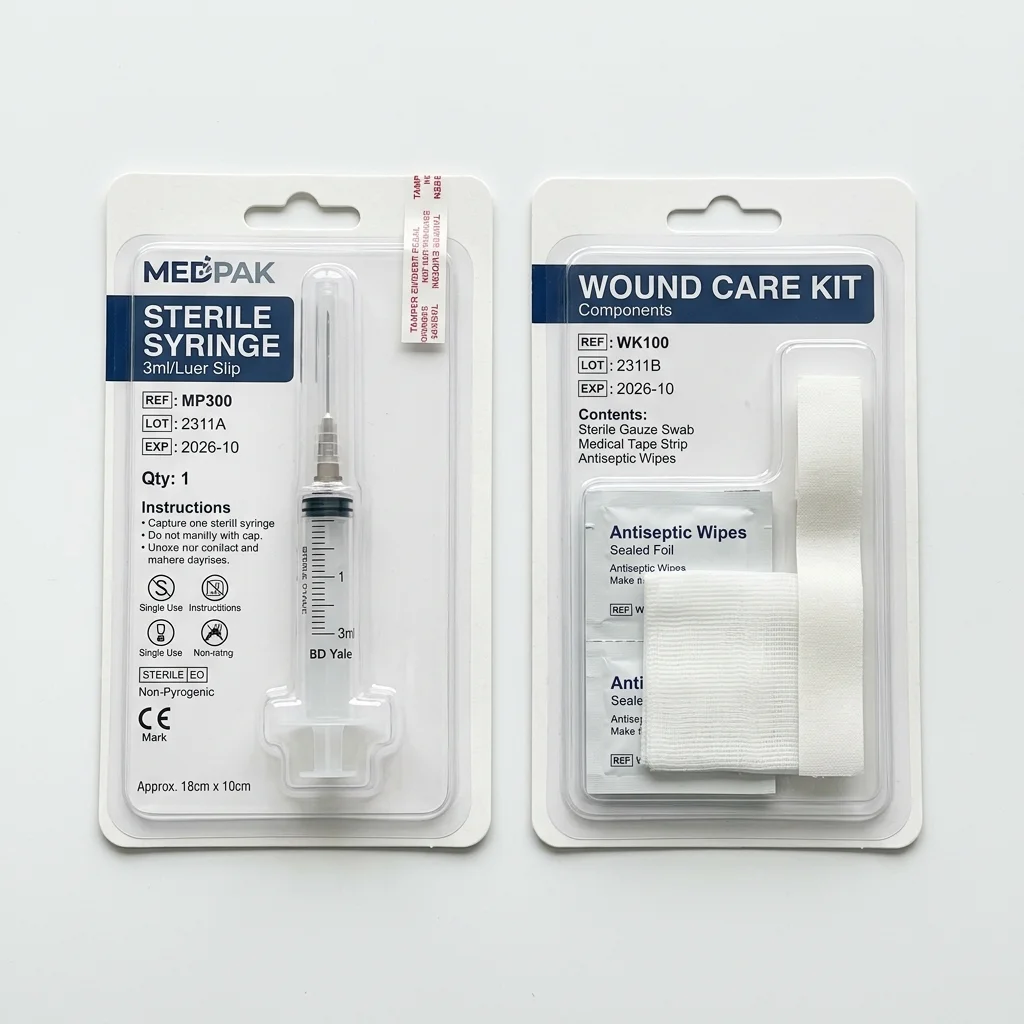
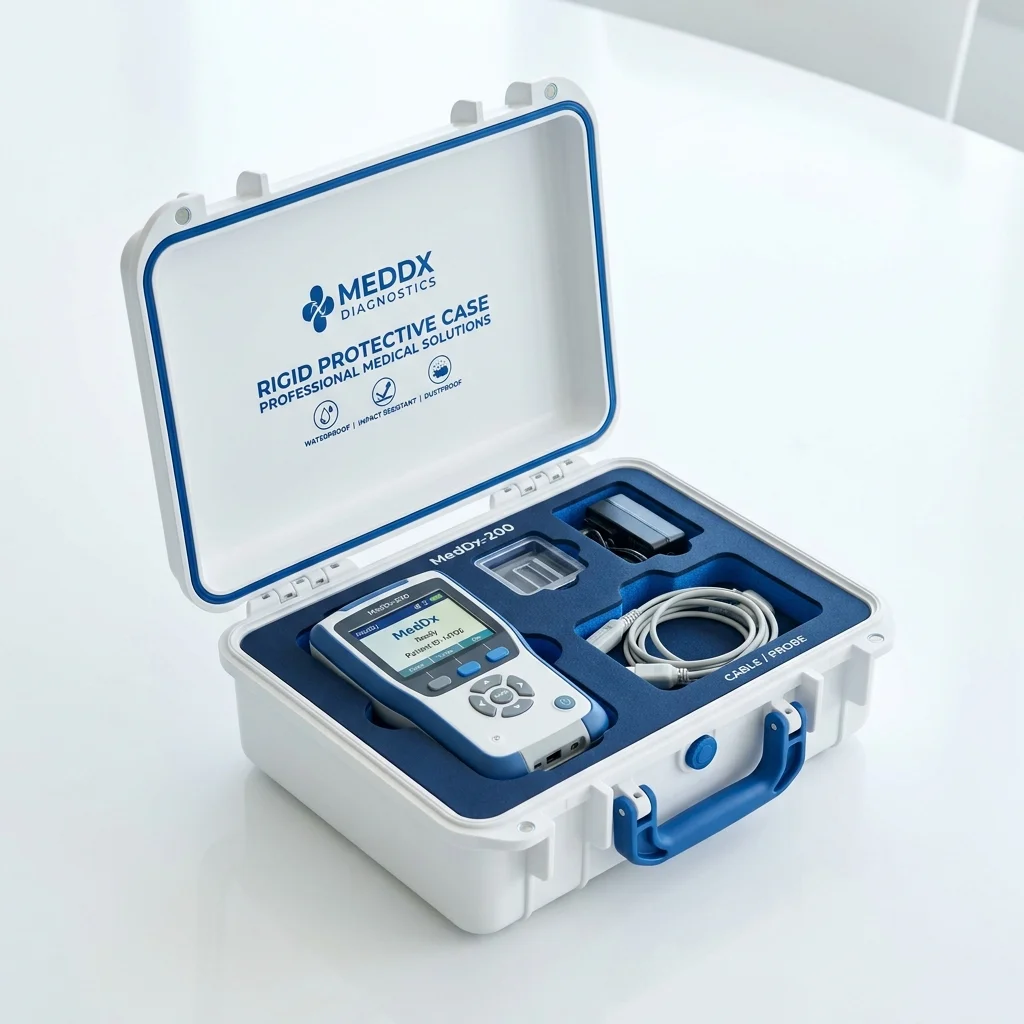
Best for: Electronic instruments, portable diagnostics, and reusable devices
Rigid Protective Case


Best for: Multi-device shipping, cold chain, and distribution packaging
Medical Corrugated Shipper
Medical Devices Packaging Requirements
Industry-specific regulations and how Cubit helps you stay compliant.
Recommended Materials for Medical Devices

Tyvek and Medical Films
- DuPont Tyvek 1073B and 1059B lidstock for EtO sterilization
- Medical-grade LDPE and HDPE film for pouch construction
- Coextruded nylon-PE films for high-barrier applications
- Breathable paper-film laminates for steam sterilization

PETG Thermoforming
- Virgin PETG sheet for optical clarity and formability
- HIPS for opaque tray applications
- Anti-static PETG for ESD-sensitive devices
- Multi-layer barrier PETG for moisture-sensitive implants

Medical-Grade Paperboard
- SBS paperboard for folding carton construction
- Clay-coated board for high-quality print reproduction
- Moisture-resistant coated board for cold chain cartons
- Recycled content board for non-sterile secondary packaging

High-Barrier Laminates
- Aluminum foil laminates for moisture and light barrier
- Metallized PET films for moderate barrier at lower cost
- EVOH-based multilayer films for oxygen barrier
- Foil-paper laminates for header bags and sachets
Brand Positioning Guide
Choose packaging that aligns with your brand's market position.
Class III High-Risk
Life-sustaining implants, cardiac devices, and neurostimulators requiring the highest levels of sterile barrier performance, traceability, and regulatory documentation. Packaging undergoes full ISO 11607 validation with accelerated aging and transit testing.
- Double sterile barrier systems with inner pouch and outer tray
- Serialized tracking with RFID and NFC authentication
- Full ISO 11607 validation package with aging studies
- Tamper-evident closures with void-pattern security labels
Class II Moderate-Risk
Surgical instruments, diagnostic equipment, and powered medical devices. The largest medical device category requiring validated sterile packaging for terminally sterilized products and regulatory-compliant cartons for non-sterile equipment.
- Thermoformed trays with Tyvek for sterile instruments
- Custom foam insert rigid cases for electronic devices
- Folding cartons with UDI panels for diagnostic kits
- Cold chain shippers for temperature-sensitive reagents
Class I Low-Risk
Bandages, examination gloves, tongue depressors, and basic medical supplies. High-volume products requiring cost-effective packaging with regulatory labeling compliance and efficient distribution packaging.
- Cost-effective folding cartons with regulatory panels
- Poly bags and header cards for individual items
- Multi-pack corrugated shippers for bulk distribution
- Simple blister cards for retail medical products
Compliance & Certifications
ISO 11607-1 and 11607-2
International standard for packaging of terminally sterilized medical devices. Part 1 covers material and sterile barrier system requirements. Part 2 covers validation of forming, sealing, and assembly processes.
FDA 21 CFR 820 / QMSR
Quality System Regulation governing medical device manufacturing including packaging design controls, process validation, and documentation requirements for the US market.
ISO 13485
Quality management system standard for medical device organizations. Ensures systematic approach to design, development, production, and packaging of medical devices.
EU MDR 2017/745
European Medical Device Regulation requiring specific labeling, traceability, and UDI implementation. Packaging must comply with Annex I general safety and performance requirements.
ASTM D4169 / ISTA Testing
Distribution testing standards validating that medical device packaging maintains product protection and sterile barrier integrity through simulated shipping and handling conditions.
ISO 14644 Cleanroom Standards
Classification system for cleanroom environments. Medical packaging operations may require ISO Class 7 or Class 8 environments depending on device classification and contamination risk.
Medical Devices Packaging Trends
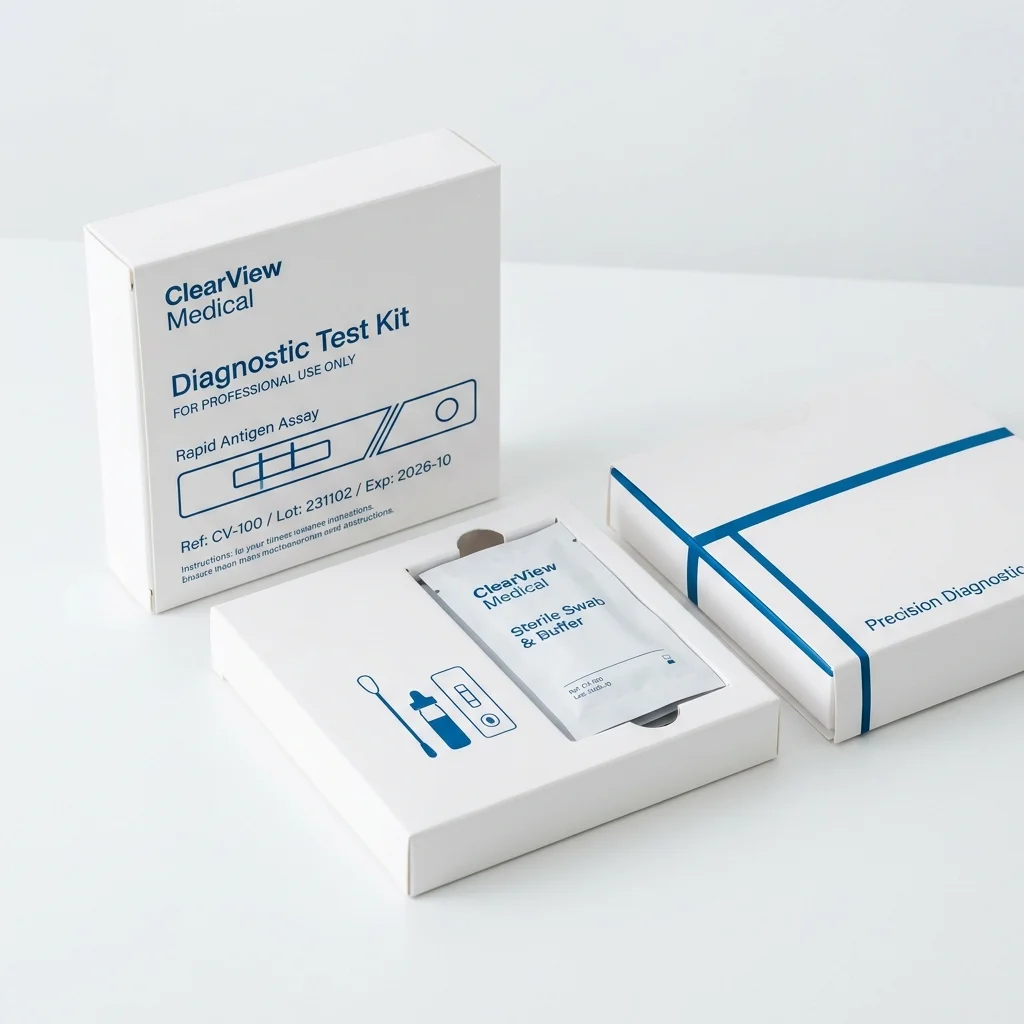
Minimalist Clinical
Ultra-clean packaging with generous white space, precise blue typography, and thin accent lines. Medical devices benefit from packaging that communicates clinical precision and professional credibility through simplicity rather than visual complexity.
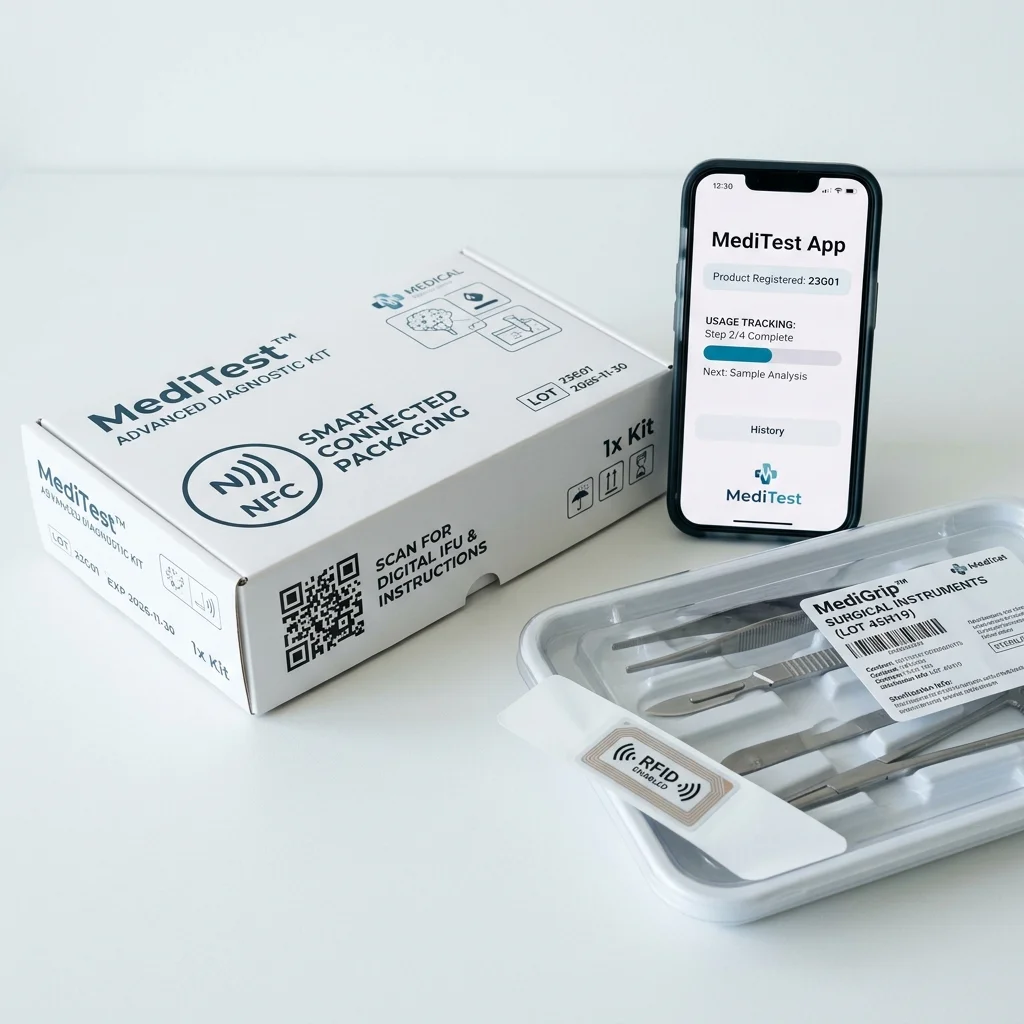
Smart Connected Packaging
Integration of NFC chips, RFID tags, and QR codes that transform packaging into data channels. Clinicians access digital IFUs, track device provenance, and register products by scanning the package instead of searching through paper inserts.

Sustainable Medical Packaging
Mono-material designs, recyclable paper-based alternatives, and right-sized formats that reduce environmental impact without compromising sterile barrier performance. Paper-based trays replacing plastic for non-sterile applications lead the shift.

Right-Sizing and Optimization
Precisely fitted packaging that eliminates excess void fill and reduces material consumption by 30-40%. Custom die-cut inserts replace loose cushioning, cutting shipping volume and waste while improving product protection through form-fitting design.
Connect with Your Customers
Add digital experiences to your packaging with QR codes and NFC technology.

UDI Barcode Integration
GS1-compliant UDI barcodes with lot, batch, and serial number encoding integrated into packaging labels. Smartphone scanning provides instant access to device registration, expiration dates, and recall status through GUDID database linking.

NFC Anti-Counterfeiting
Embedded NFC chips in premium device packaging enable tap-to-verify product authenticity. Protects high-value implants and instruments against counterfeiting while providing chain-of-custody verification from manufacturer to clinical use.

Temperature Monitoring Indicators
Time-temperature indicator labels integrated into cold chain shippers that change color irreversibly when temperature thresholds are breached. Freeze indicators, warm indicators, and cumulative time-temperature monitors protect reagents and biologics.

Tamper-Evident Security
Multi-layer packaging integrity verification: holographic security seals with void patterns, irreversible color-change indicators on sterile barriers, and serialized security labels that confirm first-open status and protect against tampering.
Eco-Friendly Options
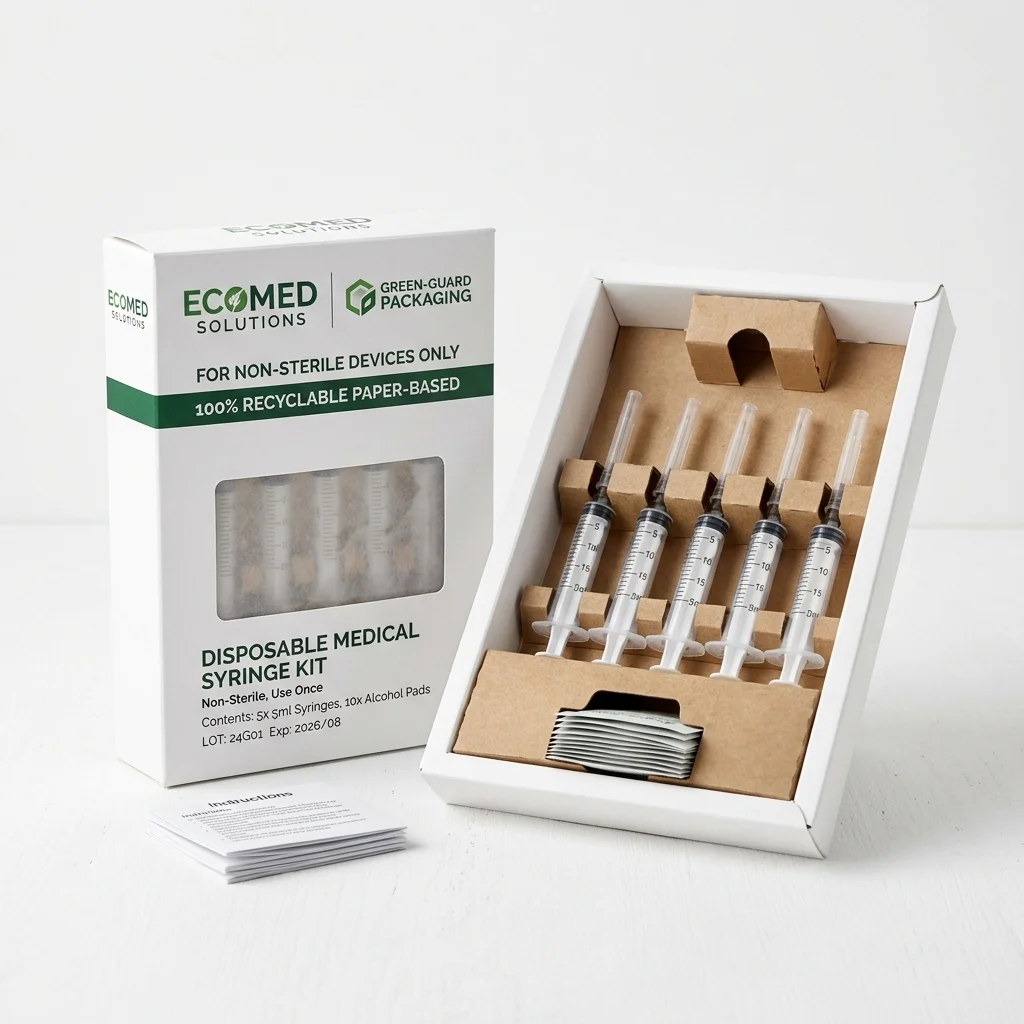
Recyclable Paper-Based Packaging
100% recyclable paperboard cartons and paper-based insert trays replacing plastic blisters for non-sterile medical devices. Maintains clinical clean appearance while enabling curbside recycling.

Right-Sized Packaging
Precisely fitted packaging that reduces material consumption by 30-40% compared to standard sizes. Custom die-cut inserts eliminate void fill while improving product protection through form-fitting design.

Mono-Material Design
Packaging systems using a single polymer family for simplified end-of-life recycling. All-PE pouch systems and all-paper secondary packaging enable single-stream recycling without material separation.

Post-Consumer Recycled Content
Secondary packaging and shippers made with PCR paperboard and recycled corrugated. Maintains clinical appearance while diverting waste from landfills. Suitable for non-sterile and distribution packaging.

Bio-Based Materials
Plant-derived polymers for thermoformed trays and films. Bio-PET and sugarcane bagasse molded fiber reduce fossil fuel dependency while meeting medical packaging performance requirements for non-sterile applications.

Carbon-Neutral Production
Manufacturing with verified carbon offsets and renewable energy sourcing. Includes carbon-neutral certification badge for packaging display, supporting medical companies ESG reporting requirements.
Medical Devices Packaging FAQ
Need Custom Medical Devices Packaging?
Our AI Packaging Consultant can analyze your product and recommend the perfect specs in seconds.